Unit | Parasite Chemotherapy
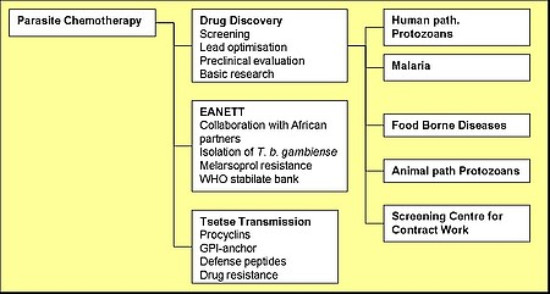
The Parasite Chemotherapy Unit (PCU) is a drug discovery centre for protozoan parasites. We have over 20 years of experience in assay development and drug efficacy testing for the causative agents of malaria (Plasmodium spp.), African sleeping sickness (Trypanosoma brucei), Chagas disease (T. cruzi), leishmaniasis (Leishmania spp.) and diarrhoea (Entamoeba and Giardia). Primary and secondary in vitro assays are combined with mouse models of disease (for African trypanosomes and malaria) and molecular approaches to drug action and resistance. The PCU works closely with product development partnerships such as the Medicines for Malaria Venture (MMV), DNDi or GALVmed, with academic partners around the world and with pharmaceutical companies. The PCU has been instrumental in the development of several clinical candidates, including fexinidazole for human African trypanosomiasis (HAT) and artefenomel for malaria.
Drug Discovery
A substantial part of the research is focused on the development of novel in vitro assays and in vivo models. Drug discovery activities are complemented by preclinical studies on drug absorption and pharmacokinetics. Molecular biology and bioinformatics approaches are also used to investigate mechanisms of drug resistance and mode of drug action. The unit has a screening mandate from the Medicines for Malaria Venture (MMV) Foundation and has become the main WHO/TDR centre for in vitro screening against protozoan parasites.
Eastern African Network for Trypanosomiasis (EANETT)
As well as working on new drugs, the group also collaborates with African partner institutes in the Eastern Africa Network for Trypanosomiasis (EANETT) in the area of sleeping sickness research and control. The Swiss Agency for Development and Cooperation (SDC) has awarded the network a second phase of support for 2004-2006. A key objective for this phase was to involve the network in international activities, with access to additional funding.
Trypanosome Transmission
Another element of the activities is to study the transmission of trypanosomes by the tsetse fly vector in order to gain more knowledge about the life cycle of the parasite and the function of certain surface proteins.

Pascal Mäser
Professor, PhD
Head of Unit
+41612848338
pascal.maeser@swisstph.ch
Selected Projects
All ProjectsLatest Publications
All PublicationsAdebayo G et al. The importance of murine models in determining in vivo pharmacokinetics, safety, and efficacy in antimalarial drug discovery. Pharmaceuticals (Basel). 2025;18(3). DOI: 10.3390/ph18030424
Beneke T et al. Leishmania mexicana pathogenicity requires flagellar assembly but not motility. Virulence. 2025;16(1):2521478. DOI: 10.1080/21505594.2025.2521478
Dam J et al. Cell-based progression of spiroindoline phenotypic hits leads to the identification of compounds with diverging parasitological profiles against the human malaria parasite Plasmodium falciparum. J Med Chem. 2025;68(10):10156-10172. DOI: 10.1021/acs.jmedchem.5c00302
Dupouy B et al. Click coupling of flavylium dyes with plasmodione analogues: towards new redox-sensitive pro-fluorophores. Chemistry. 2025;31(6):e202403691. DOI: 10.1002/chem.202403691
Dupouy B et al. Synthesis of 1,2,3-triazole-methyl-menadione derivatives: evaluation of electrochemical and antiparasitic properties against two blood-dwelling parasites. ChemMedChem. 2025;20(6):e202400731. DOI: 10.1002/cmdc.202400731

 Andreia Albuquerque Wendt
Andreia Albuquerque Wendt
 Christin Gumpp
Christin Gumpp
 Malenka Kunz
Malenka Kunz
 Matthias Rottmann
Matthias Rottmann