
Preclinical research in infection biology
Swiss TPH plays a key role in developing vaccines, drugs and diagnostic tests for various pathogens through innovative preclinical research. This research includes the establishment and maintenance of different parasite life cycles and the development and application of in vitro, in vivo and bioanalytical assays. We also evaluate the pharmacokinetic properties of drug candidates.
Our preclinical research is closely linked to both basic infection biology research and clinical research, bridging the gap between fundamental discoveries and the development of new tools and product candidates ready for clinical validation.
Key projects
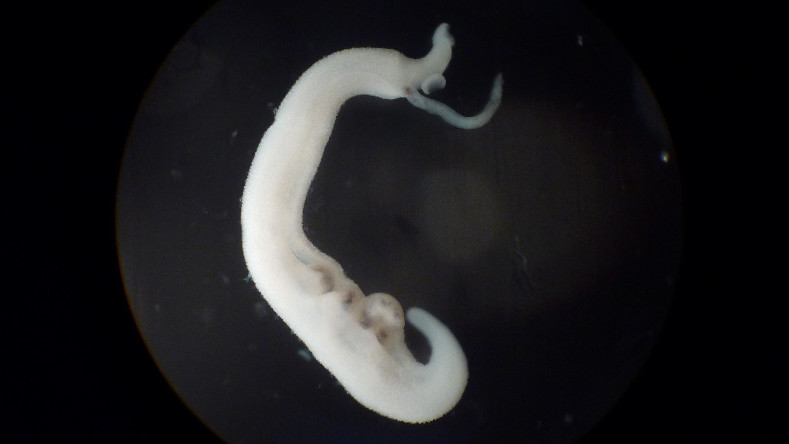
Optimisation of antischistosomal chemotypes
Drug-resistant schistosomiasis is an increasing threat and new drugs are urgently needed for integrated control programmes. We aim to identify one or more antischistosomal drug candidates that are effective against all stages of the parasite and have a novel mechanism of action. To achieve this goal, we will optimise four promising and structurally diverse antischistosomal chemotypes.
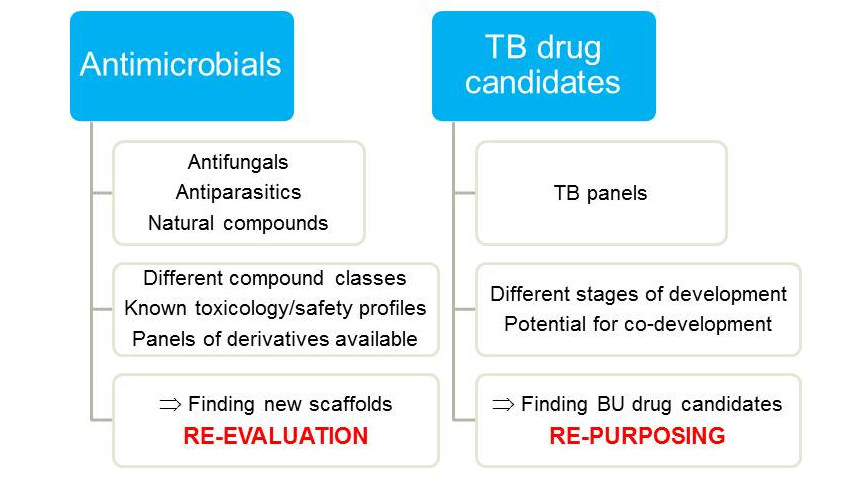
Buruli ulcer - drug screening
Re-evaluation and repurposing of antimicrobials is an effective strategy for the development of new treatment regimens for neglected tropical diseases. Given the very limited financial resources available, repurposing of tuberculosis drug development candidates is a particularly attractive approach for Buruli ulcer disease. Although most tuberculosis drug scaffolds show no or limited activity against M. ulcerans, we have identified advanced tuberculosis drug candidates suitable for co-development against both tuberculosis and Buruli ulcer.