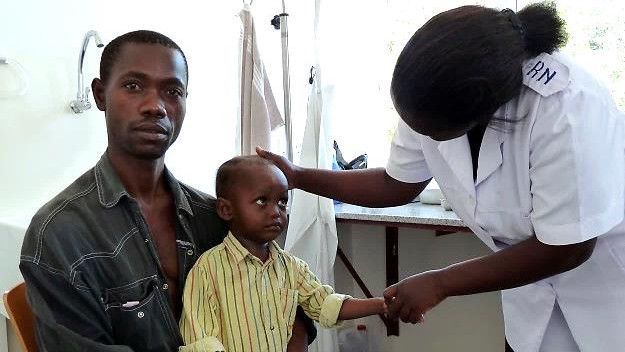
Diagnostics, Vector Control, Vaccines and New Drugs
We develop, validate and apply drugs, diagnostics, vaccines, vector control measures and computational tools to improve global health.
At Swiss TPH, we conduct product-oriented research and contribute to the entire value chain of related research and development (R&D) processes. We undertake preclinical and clinical product development of drugs, diagnostics and vaccines for poverty-related infectious diseases. Swiss TPH has a proven track record in the design, conduct and monitoring of clinical trials for vaccine candidates, novel diagnostic tools and new or repurposed drugs against parasites, viral and bacterial pathogens and for the establishment of vector control measures. Swiss TPH promotes the clinical development and post-approval validation of newly developed vaccines, drugs, diagnostic tests and vector control tools.