Malaria Gene Regulation
Malaria is a devastating infectious disease caused by protozoan parasites of the genus Plasmodium that are transmitted to humans via the bite of infected female Anopheles mosquitoes. P. falciparum causes the most severe form of malaria and is responsible for over 200 million clinical cases and 500’000 deaths each year.
Our research focus
We study the biology of P. falciparum asexual and sexual blood stage parasites at the molecular and cellular level. Asexual parasites undergo repeated rounds of red blood cell (RBC) invasion and intracellular replication, which is responsible for all malaria disease manifestations. Sexual parasites, i.e. female and male gametocytes, are the only forms of the parasite able to infect the mosquito vector and are hence responsible for malaria transmission. Gametocytes are formed at low frequency from asexual parasites via an epigenetic switch that activates expression of PfAP2-G. The transcription factor PfAP2-G initiates a transcriptional programme driving parasites irreversibly into the sexual differentiation pathway. These cells then develop over the course of 10-12 days into fully differentiated transmissible female or male stage V gametocytes.
Research on asexual parasites: antigenic variation and replication
Our research on asexual parasites focuses on the molecular mechanisms and players involved in regulating antigenic variation and parasite replication. Antigenic variation describes the clonally variant expression of surface antigens. In P. falciparum, antigenic variation is based on epigenetic control mechanisms that facilitate switches in the mutually exclusive expression of one out of 60 var gene paralogs, each of which encodes a different PfEMP1 surface antigen. Parasite replication occurs via schizogony, a specialised mode of cell division. During schizogony, 4-5 consecutive cycles of genome replication and nuclear division create a multinucleated cell that then undergoes a single cytokinesis event, resulting in the egress of 20-30 invasive merozoites from the infected RBC.
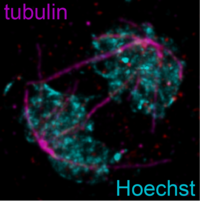
Research on sexual parasites: mechanisms of underlying gametocytogenesis
Our research on gametocytes covers the entire process of gametocytogenesis. We are interested in understanding the mechanisms that (1) control sexual commitment and conversion, i.e. the silencing and activation of the pfap2-g locus and the immediate consequences of PfAP2-G expression; (2) take place during the earliest phase of sexual differentiation, where sex-specific features are not yet readily discernible; and (3) are linked to the sex-specific maturation of gametocytes.
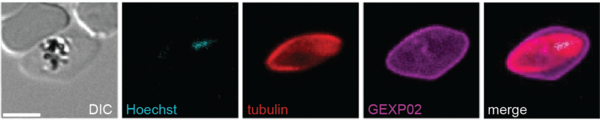
Molecular regulators and experimental approaches
The targets of our investigations are most often regulatory factors – such as epigenetic readers/writers, transcription factors and kinases – that are known or suspected to play essential roles in these processes, or that we identified experimentally. We study these factors in detail at the functional level using CRISPR/Cas9-based engineering of parasite mutants and their subsequent phenotyping by growth assays, fluorescence microscopy, flow cytometry, transcriptomics, proteomics and other methods
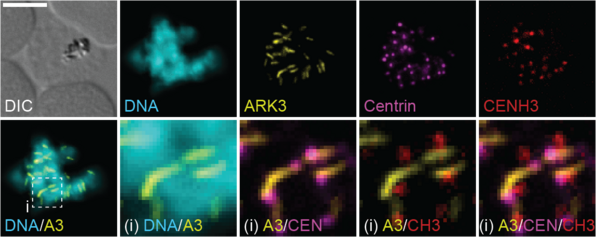
Methodological innovation
Lastly, our lab is interested in developing improved or new methodology for malaria research. We for instance engineered so called “inducible gametocyte producer” lines (3D7/iGP, NF54/iGP) that allow for the routine mass production of synchronous gametocytes. This had previously been extremely difficult to achieve due to the low sexual conversion rates usually observed in parasite in vitro cultures. These iGP lines have been a game changer for gametocyte research and are now used by us and many other labs to study various aspects of gametocyte biology at much greater pace and depth. In addition, together with the labs of Nicolas Brancucci and Matthias Rottmann (both at Swiss TPH), we developed these iGP lines further into a highly suitable tool for the discovery and preclinical development of malaria transmission blocking drugs.
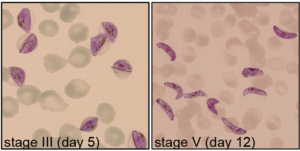
Selected projects
Exploring early sexual differentiation in the malaria parasite
The SNSF-funded research project focuses on gametocytes, the transmissible forms of the malaria parasite Plasmodium falciparum. The team, together with external collaborators, will apply CRISPR/Cas9 gene editing, super-resolution microscopy techniques and various –omics approaches to study the early phase of gametocyte differentiation and the mechanisms underlying gametocyte sex determination. With this project, the researchers aim to generate novel insight into the molecular and cellular events driving gametocytogenesis, offering potential avenues for the development of urgently needed new interventions for malaria elimination. Read more
Partners
Nicolas Brancucci (Swiss TPH, Switzerland)
Matthias Rottmann and Pascal Maeser (Swiss TPH, Switzerland)
Alexander Schmidt (Biozentrum, University of Basel, Switzerland)
Matthias Marti (University of Zurich, Switzerland)
Mathieu Brochet (University of Geneva, Switzerland)
Richard Bartfai (Radboud University, The Netherlands)
Artur Scherf (Institut Pasteur, France)
Michael Filarsky (University of Tübingen, Germany)
Zbynek Bozdech (School of Biological Sciences, Nanyang Technological University, Singapore)
Peter Preiser (School of Biological Sciences, Nanyang Technological University, Singapore)
James McCarthy and Matt Dixon (The Walter and Eliza Hall Institute of Medical Research, Australia)
Krishanpal Karmodiya (Indian Institute of Science Education and Research Pune, India)
Daniel Baeschlin (FAST Lab, Novartis Basel, Switzerland)
Former Postdocs and PhD students
Eilidh Carrington
Elvira Carrio
Michael Filarsky
Nicolas Brancucci
Christian Flueck
Beatriz Graca
Matthias Wyss
Eva Hitz
Travis Basson
Hai Bui
Nicole Bertschi
Sophie Oehring
Kathrin Witmer


 Daniel Ballmer
Daniel Ballmer
 Patricia Landeau Millan
Patricia Landeau Millan
 Igor Niederwieser
Igor Niederwieser
 Till Voss
Till Voss
 Matthias Wyss
Matthias Wyss