Milestone towards eliminating sleeping sickness
04.03.2026
The European Medicines Agency has issued a positive opinion for Acoziborole Winthrop, a single-dose oral treatment for T.b. gambiense, the most common form of sleeping sickness. Developed over two decades by the Drugs for Neglected Diseases initiative (DNDi), Sanofi and partners, it marks a significant milestone in the fight against a neglected disease affecting some of the world's most vulnerable populations. Swiss TPH was among the partners contributing to its development.
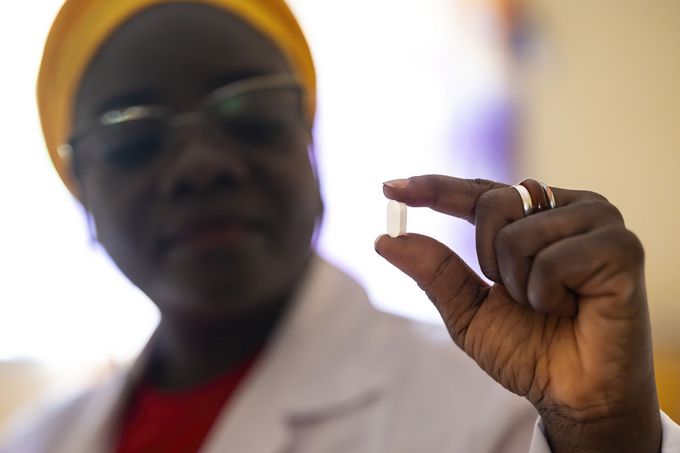
Photo: Brent Stirton/Getty Images for DNDi
Human African trypanosomiasis, also known as sleeping sickness, a neglected tropical disease, is usually fatal without treatment. Transmitted by the tsetse fly across sub-Saharan Africa, it puts around 65 million people at risk and in its later stages, causes severe neurological symptoms before leading to death. Until now, treatment required multi-day regimens of oral medication or injections.
Acoziborole Winthrop is given as a single dose in three tablets and is indicated for adults and adolescents aged 12 and older, covering both early and advanced stages of gambiense sleeping sickness. The positive opinion is based on a Phase II/III clinical trial published in The Lancet Infectious Diseases, which demonstrated success rates of up to 96% at 18 months across both stages of the disease. The success of acoziborole is a testament to more than two decades of commitment from partners.
Swiss TPH’s contribution to Acoziborole Winthrop spans two decades of collaboration with DNDi, Sanofi, and others – from early-stage screening and preclinical evaluation of the compound class that gave rise to acoziborole, to monitoring the pivotal Phase III trial in the Democratic Republic of Congo, the country most affected by the disease. This is part of a longer research commitment: for decades, Swiss TPH has been at the forefront of sleeping sickness research – from pioneering drug efficacy testing in the laboratory to conducting the first modern clinical trial on the disease in the Democratic Republic of the Congo (DRC) in the late 1990s.
The decision is expected to support regulatory approval in the DRC and inform a revision of WHO treatment guidelines, broadening access across Central and West Africa. Sanofi will donate the medicine to the WHO, making it available free of charge to patients, bringing the WHO's goal of eliminating sleeping sickness by 2030 within reach.
Contact for media requests: communications@swisstph.ch
Stay connected to global health
Subscribe to the Swiss TPH newsletter for updates on research, projects, education, jobs and events.



