Unit | Clinical Research
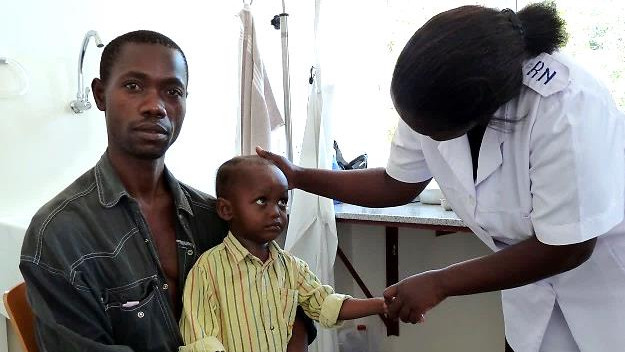
The Clinical Research Unit (CRU) at Swiss TPH designs, conducts, and reports on clinical research studies, primarily in and for low- and middle-income countries, with a focus on vulnerable populations.
In collaboration with international partner institutions, we conduct high-quality scientific studies in the areas of tuberculosis, HIV/AIDS, febrile illnesses, neglected tropical diseases, non-communicable chronic diseases, and mental health. The goal of our work is to gain knowledge about novel clinical tools or interventions, share our findings with the scientific community and the public, inform health policy, and translate knowledge into improved health. We typically work in multidisciplinary and transdisciplinary projects in which building research capacity plays a central role. We provide teaching, training and mentoring for students and young scientists at Swiss TPH and at our partner organizations around the globe.

Klaus Reither
PD Dr. med. et phil.
Head of Unit
+41612848967
klaus.reither@swisstph.ch
Main CRU Activities
Latest Publications
All PublicationsBrown J.A et al. Cohort profile: the Viral load Cohort North-East Lesotho (VICONEL) from 2016 to 2023 - cohort description, test volumes, predictors of viraemia and the road ahead. BMJ Open. 2025;15(2):e085404. DOI: 10.1136/bmjopen-2024-085404
Chakhaia T et al. Lack of weight gain and increased mortality during and after treatment among adults with drug-resistant tuberculosis: a retrospective cohort study in Georgia, 2009-2020. ERJ Open Res. 2025;11(3):00839-2024. DOI: 10.1183/23120541.00839-2024
Collins J.M et al. A plasma metabolic signature to diagnose pulmonary tuberculosis and monitor treatment response. J Infect Dis. 2025(in press). DOI: 10.1093/infdis/jiaf240
Gils T et al. A prospective evaluation of the diagnostic accuracy of the point-of-care VISITECT CD4 Advanced Disease test in seven countries. J Infect Dis. 2025;231(1):e82-e90. DOI: 10.1093/infdis/jiae374
Goig G.A et al. Transmission as a key driver of resistance to the new tuberculosis drugs. N Engl J Med. 2025;392(1):97-99. DOI: 10.1056/NEJMc2404644
