First Malaria Vaccine Recommended for Children by the WHO
07.10.2021
Yesterday, the World Health Organization (WHO) recommended the widespread use of the malaria vaccine RTS,S/AS01, debuting a tool that could save the lives of tens of thousands of children in sub-Saharan Africa each year. The vaccine is not just a first for malaria — it is the first developed for any parasitic disease. This day was made possible through decades-long research and teaching commitments to advance malaria eradication.
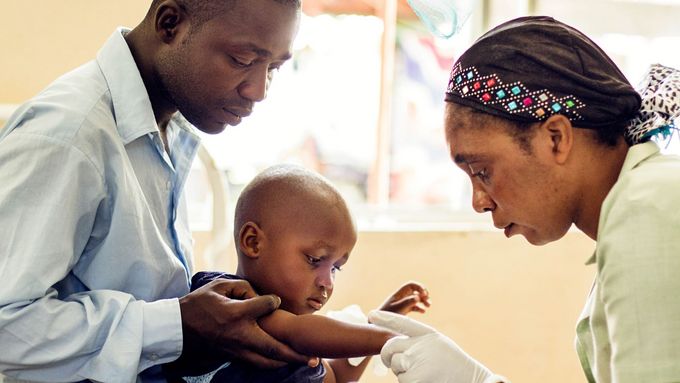
Malaria remains a primary cause of childhood illness and death in sub-Saharan Africa. WHO’s recommendation offers a glimmer of hope for the continent. (Photo: Matthis Kleeb, Swiss TPH)
The World Health Organization (WHO) endorsed the widespread use of the malaria vaccine RTS,S/AS01, marking the first step in a process that should lead to broad distribution in low- and middle-income countries. The vaccine assists a child’s immune system to prevent an infection by Plasmodium falciparum, the deadliest of the five malaria pathogens and the most common in Africa.
Swiss TPH has played a leading role in this day coming to fruition thanks to decades of research and development of new malaria vaccines. Researchers at Swiss TPH contributed across the entire development pathway from discovery to preclinical studies, and through to human clinical testing in both early and late studies with many African collaborators.
“Today is a historic moment in the fight against this age-old disease,” said Christian Lengeler, a malaria epidemiologist at Swiss TPH and President of the Swiss Malaria Group. “With a moderate efficacy of 30% of severe malaria cases, the new vaccine still makes a very important public health contribution because this disease is so common and negatively affects the lives of millions of children in Africa.”
First generation of malaria vaccines
For over 30 years, Swiss TPH has contributed to the RTS,S vaccine development through a multitude of research, validation and testing projects. Under the leadership of Claudia Daubenberger, researchers have worked to understand the response of the human immune system to the vaccine. RTS,S, produced by GlaxoSmithKline (GSK), belongs to a first generation of malaria vaccines.
"Depending on the country and malaria transmission, and in addition to other preventive measures such as mosquito nets and early diagnosis and treatment, the vaccine is of great importance," said Marcel Tanner, Director Emeritus of Swiss TPH. Tanner was a central figure in the development and clinical validation of the vaccine in all the clinical phases. Together with Tanzanian partners, he set up state of the art trial infrastructure in Bagamoyo, Tanzania and consolidated the partnership with African stakeholders in order to ensure that the vaccine could be organised and evaluated in the Phase III clinical trials in 11 trial centres across the African continent. This ensured that the pilot implementations leading to the WHO recommendation could be undertaken.
Disease and vaccine modelling at Swiss TPH
For 13 years, the Disease Modelling Unit at Swiss TPH contributed through building models of the likely impact and cost-effectiveness of the vaccine. “We’re very proud that our modelling work, in collaboration with Imperial College London and PATH, could contribute to the important WHO recommendation yesterday,” said Melissa Penny, lead of the mathematical modelling team on the project. “Our work has helped to show the impact of the vaccine on saving lives and averting disease. Modelling was an important part of this recommendation by estimating the effect of the vaccine beyond what was shown in the trial studies.”
In their models, scientists took into account demographic and epidemiological data, vaccination coverage rates and people's access to healthcare. The models found RTS,S to have a high impact in countries with moderate to high malaria transmission. The models have supported key organisations such as the WHO and GAVI the Vaccine Alliance for their decision-making on the vaccine.
Future of malaria vaccines
RTS,S is the first first malaria vaccine recommended by WHO for broad use among young children in Africa and in other areas with moderate to high transmission of P. falciparum malaria. Next steps will include funding decisions from the global health community for broader rollout, and country decision-making on whether to adopt the vaccine as part of national malaria control strategies.
With mRNA vaccination developed in the context of the COVID-19 pandemic, new technologies are now also available for malaria vaccine development in the future. "We are only at the beginning of an important development and the long-held dream of one day being able to effectively combat and eliminate malaria," said Tanner.
Stay connected
Subscribe to our newsletter and get all the latest research news, project updates, course and event listings from Swiss TPH.

