Human African Tryponosomiasis
African sleeping sickness remains a deadly disease, but it will take a concerted effort to eradicate it. For decades, Swiss TPH has been at the forefront of developing innovative ways to combat the deadly parasites responsible for the disease. Over the past decade, our researchers have identified dozens of highly potent compounds - some with entirely new properties. The most promising of these are now undergoing further preclinical evaluation, bringing us one step closer to effective new treatments.
From bench to bedside: overcoming challenges
While cutting-edge drug research continues in the laboratory, Swiss TPH teams are also working on the ground in some of the world’s most remote regions. Conducting clinical research in these areas means facing extreme challenges - difficult travel, scarce medical resources, and complex logistical hurdles. Despite these obstacles, our commitment to the highest clinical standards ensures that every trial we conduct brings us closer to better treatments and, ultimately, disease elimination.
A future without West African sleeping sickness?
Swiss TPH experts have evaluated and modelled multiple strategies to stop the spread of West African sleeping sickness. Their findings reveal that a comprehensive approach is essential: in addition to introducing new drugs, targeted elimination of tsetse fly populations, improved diagnostics, and strengthened disease surveillance are all critical to achieving a lasting impact. With continued research, field work, and global collaboration, the eradication of African sleeping sickness is within reach.
The disease
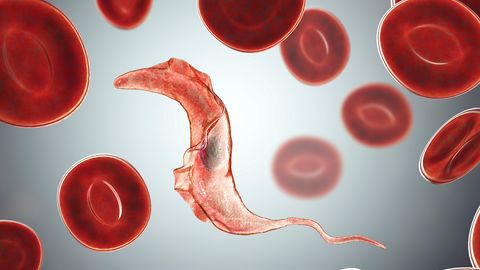
West African sleeping sickness is a prime example of a neglected tropical disease (NTD). It occurs in the most remote regions of sub-Saharan Africa and primarily affects the poorest populations. The protozoan parasite that causes the disease, Trypanosoma brucei gambiense, is transmitted to humans by the tsetse fly. Without treatment, HAT is generally fatal.
After the initial phase of non-specific pain, fever, rash and swelling, the parasite crosses the blood-brain barrier and attacks the brain. Severe psychiatric disorders, sleeplessness and impaired speech, body coordination and feeding eventually lead to death. The WHO has set a target of eliminating the disease as a public health problem by 2020 and stopping transmission (zero cases) by 2030.

Selected projects

Major milestone in the fight against sleeping sickness
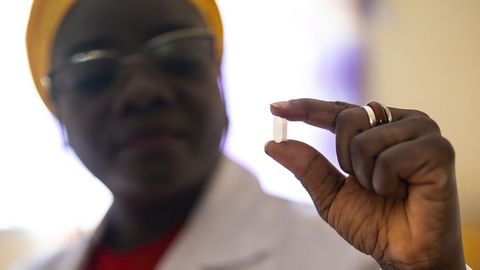
In December 2023, the European Medicines Agency (EMA) issued a positive opinion for fexinidazole for the treatment of T.b. rhodesiense sleeping sickness, an acute and deadly form of this neglected tropical disease (NTD) found in eastern and southern Africa. The decision followed a clinical trial led by the HAT-r-ACC consortium, including Swiss TPH. This marks a significant breakthrough in combating acute sleeping sickness, improving access to treatment and saving lives in affected regions. Read more

Fexinidazole: a breakthrough oral treatment for sleeping sickness
In November 2018, the European Medicines Agency’s Committee for Medicinal Products for Human Use (CHMP) approved fexinidazole, the first all-oral treatment effective for both stages of sleeping sickness. This milestone followed clinical trials led by DNDi and an application by Sanofi, with Swiss TPH playing a key role in the drug's development. Fexinidazole treats both the early stage of infection and the advanced stage, when the parasites cross the blood-brain barrier and cause neuropsychiatric symptoms. The approval is a major step forward in the fight against the NTD. Read more

Drug discovery in collaboration with DNDi
The Drugs for Neglected Diseases initiative (DNDi) is a non-profit research organisation dedicated to discovering, developing, and delivering affordable, patient-friendly treatments for neglected patients worldwide. Swiss TPH partners with DNDi in drug discovery, providing in vitro assays to test parasite responses, conducting cytotoxicity assessments and developing rodent infection models for African trypanosomes. This collaboration accelerates the search for safe, effective treatments for neglected tropical diseases, bringing hope to affected communities.